AP26194471 – Metagenomic and metatranscriptomic studies of extreme ecosystems to explore the unique metabolic capabilities of microorganisms in green biofuel technologies
Objective of the project – to explore metagenomic and metatranscriptomic data from extreme ecosystems to identify and expand a collection of microorganisms with unique metabolic capabilities, forming a foundation for the development of innovative and environmentally sustainable biofuel technologies.
Relevance: The relevance of this project is driven by the vast yet poorly explored diversity of microorganisms and their untapped metabolic potential for bioenergy applications. Despite advances in microbiology, most microorganisms remain unknown, especially those from extreme ecosystems such as hot springs, saline environments, and deep anaerobic habitats. These environments host unique microbial communities with adaptive metabolic capabilities that can provide innovative solutions for the development of green biofuels. Traditional research methods are limited in their ability to capture the full spectrum of microbial diversity, making advanced approaches like metagenomics and metatranscriptomics essential. These modern techniques enable access to hidden genetic and functional information, allowing the identification of key metabolic pathways for biohydrogen, biobutanol, and biodiesel production. Therefore, the project is highly relevant as it supports the transition to sustainable energy by unlocking microbial resources and contributing to the development of environmentally friendly biofuel technologies.
Scientific supervisor: Ph.D., Associate Professor, Kakimova Ardak Bolatovna
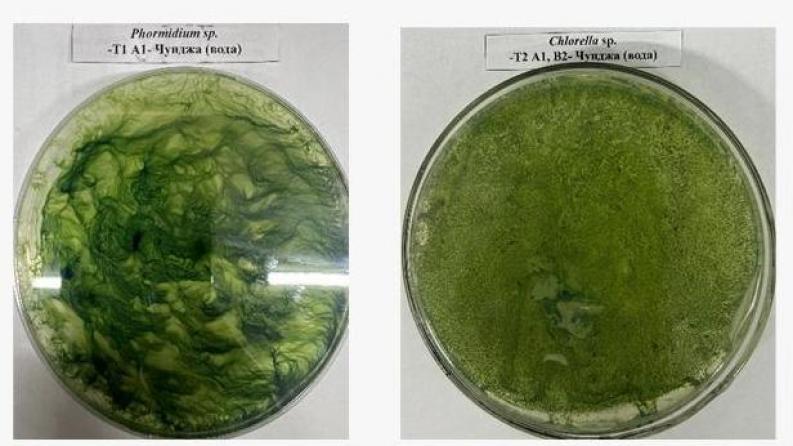
Expected and achieved results: The project has successfully completed large-scale field studies across multiple extreme ecological regions of Kazakhstan, including Almaty, East Kazakhstan, Kyzylorda, West Kazakhstan, and Pavlodar regions. Soil and water samples were collected from diverse environments such as geothermal springs, saline soils, desert areas, and mineralized water bodies, ensuring a representative dataset of extremophilic microbial communities. All sampling procedures were conducted in compliance with biosafety standards, including sterile collection, GPS tracking, and proper documentation and transportation. High-quality DNA was extracted from the collected samples using optimized protocols, providing a reliable basis for further metagenomic analysis. In the second stage, a comprehensive metagenomic analysis was carried out on selected samples, each processed with biological replicates to ensure statistical reliability. The samples were sequenced using next-generation sequencing (NGS) technologies, followed by advanced bioinformatics processing, including filtering, annotation, and taxonomic profiling. The results revealed that bacterial communities dominate across all samples, with a significant presence of archaea adapted to extreme conditions such as high salinity and temperature fluctuations. A wide diversity of microbial genera was identified, including Clostridium, Acetobacterium, Desulfovibrio, Halomonas, Synechococcus, and Nostoc, indicating a broad range of metabolic capabilities. Functional analysis detected key genes associated with hydrogen metabolism, including [FeFe] and [NiFe] hydrogenases and their maturation genes, confirming the potential for biological hydrogen production. Additionally, nitrogenase gene clusters were identified, suggesting dual pathways for hydrogen generation under anaerobic conditions. The study also identified genes involved in lipid biosynthesis and fatty acid metabolism, indicating the presence of microorganisms with high lipid accumulation potential suitable for biodiesel production. Genes related to butanol and ABE fermentation pathways were detected, demonstrating the capability of microbial consortia to produce biofuels such as biobutanol, acetone, and ethanol. Furthermore, a wide range of enzymes responsible for plant biomass degradation, including cellulases and lignin-degrading enzymes, were identified, confirming the ability of these communities to convert organic substrates. The presence of stress-resistance genes related to salinity, temperature, and oxidative stress highlights the strong adaptive capacity of the identified microorganisms. Based on the obtained results, priority samples have been selected for further isolation of pure cultures with high biotechnological potential. Overall, the project has generated a valuable scientific dataset and established a solid foundation for developing sustainable biofuel technologies based on extremophilic microorganisms.
